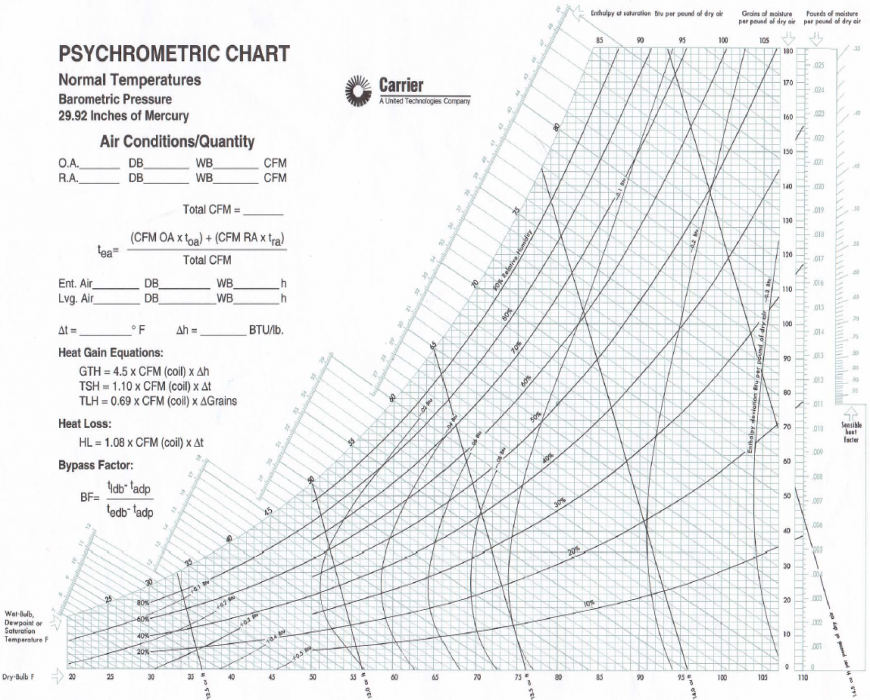
Its specific volume is increased, in accordance with the law of gases, in direct proportion to the increase of absolute temperature, while its density is changed in an inverse proportion, as shown in Fig. This is the well-known temperature-pressure curve of steam.ħ When the temperature of a definite weight of saturated vapor is increased isobarometrically, it is said to be superheated. In the same manner, an unlimited amount of a gas, such as air, could be added isothermally to a cubic foot of water vapor without affecting its condition of saturation, giving a combined pressure equal to the gas pressure plus the vapor pressure.Ħ The established temperature-pressure relationship of saturated water vapor is shown by curve (1) on the charts, Figs. But if, on the other hand, it is attempted to include isothermally an additional amount of either of the saturated vapors, a corresponding condensation of the particular vapor added would result. Similarly, an equal volume of a third saturated vapor might be added without affecting the other two. cent., having as a total pressure the sum of the two separate saturated vapor pressures, or 2457.6 mm. of the mixture saturated with both water vapor and alcohol vapor at 100 deg. cent, having a vapor pressure of 1697.6 mm., and add isothermally to this 1 cu. 
saturated with vapor of alcohol at 100 deg. This vapor, according to Dalton's law, is capable of exerting a certain maximum vapor pressure dependent entirely on its temperature and regardless of the presence of other gases or vapors. VAPOR PRESSURE AND LAW OF PARTIAL PRESSURESĥ Water vapor exists in the air purely as a mixture in relation to its other elements. At the same time, sufficient error has been shown in previous steam data, especially at atmospheric temperatures, to warrant the revision of all calculations based thereon. Recent investigators have determined the most important properties of water vapor with final accuracy. .png)
Accepted data used in psychrometric calculations are based largely on empirical formulae, which are incorrect as well as limited in their range. While air conditioning has never been properly applied to coal mines, the author is convinced that if this were made compulsory, the greater number of mine explosions would be prevented.Ģ Although of so much practical as well as scientific importance the laws governing many of the phenomena of atmospheric moisture are but partially understood, while the present engineering data pertaining thereto are both inaccurate and incomplete. In many other industries, such as lithographing, the manufacture of candy, bread, high explosives and photographic films, and the drying and preparing of delicate hygroscopic materials, such as macaroni and tobacco, the question of humidity is equally important. When applied to the blast furnace, it has increased the net profit in the production of pig iron from $0.50 to $0.70 per ton, and in the textile mill it has increased the output from 5 to 15 per cent, at the same time greatly improving the quality and the hygienic conditions surrounding the operative. The application of this new art to many varied industries has been demonstrated to be of greatest economic importance. A specialized engineering field has recently developed, technically known as air conditioning, or the artificial regulation of atmospheric moisture.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
